- Product Details
Keywords
- research chemical
- similar to GLYX-13
- for major depressive disorder (MDD)
Quick Details
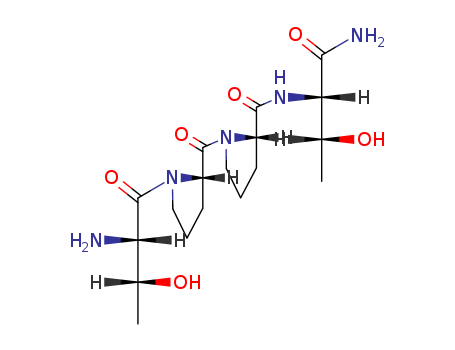
- ProName: NRX-1074
- CasNo: 117928-94-6
- Application: treatment of major depressive disorder...
- ProductionCapacity: Metric Ton/Day
- Purity: 98%
- LimitNum: 0 Metric Ton
Superiority
| 77472-70-9 | phenylpiracetam | |
| 110958-19-5 | Fasoracetam | |
| 15180-02-6 | Amfonelic acid | |
| 68497-62-1 | Pramiracetam | |
| 116041-13-5 | Nebracetam | |
| 77191-36-7 | Nefiracetam | |
| 62613-82-5 | Oxiracetam | |
| 72432-10-1 | aniracetam | |
| 157115-85-0 | Noopept | |
| 59776-89-5 | Piracetam hydrazide | |
| 51352-87-5 | PRL-8-53 | |
| 22503-72-6 | IDRA-21 | |
| 1270138-40-3 | NSI-189 | |
| 272786-64-8 | Unifiram | |
| 314728-85-3 | Sunifiram | |
| 63547-13-7 | Adrafinil | |
| 33818-15-4 | Citicoline Sodium | |
| 987-78-0 | Citicoline | |
| 28319-77-9 | Alpha-GPC | |
| 120786-18-7 | Huperzine A | |
| 42971-09-5 | Vinpocetine | |
| 34271-54-0 | L-theanine | |
| 38183-03-8 | 7, 8 dihydroxy flavonoids | |
| 1689-64-1 | 9-fluorenol | |
| 127464-43-1 |
Mexidol |
Details
NRX-1074 is a novel modulator of the NMDA receptor. It is a second-generation follow-on to Naurex’s lead compound, GLYX-13. NRX-1074 is similar to GLYX-13, but is orally active and significantly more potent. The drug is in Phase 2 clinical development for the treatment of major depressive disorder (MDD).
The objectives of the Phase 2 study now underway are to evaluate the safety and efficacy of a single dose of NRX-1074 administered to subjects with MDD. This Phase 2 study follows successful completion of a randomized, placebo-controlled Phase 1 study that assessed the safety of the compound in normal volunteers. In this study, NRX-1074 was well-tolerated, with no signs of the schizophrenia-like psychotomimetic effects associated with NMDA receptor antagonists, such as ketamine.
NRX-1074 has mechanistic activity similar to that of GLYX-13. In multiple nonclinical studies, NRX-1074 has shown rapid onset and long duration of antidepressant-like effects with good safety and no signs of CNS-related side effects.